Table of Contents
I. Overview
I. Overview
- EWB Evaluation
- WHO Specifications
- Pulmonary Biomechanics
- Review Criteria
- Criteria Scoring
|
To the right, you will find EWB's evaluation of open-source ventilator projects. Please read our take on invasive ventilators in the context of COVID-19 response before examining the rest of the information provided.
|
|
Pulmonary Biomechanics
|
Dr. M has 10 years of experience studying the lungs through a biomedical engineering lens. Most recently (2016-2019), she trained in pulmonary biomechanics on the Anschutz Medical Campus, creating patient-specific predictive models to try to identify safe ventilation in animals with ARDS.
|
In an attempt to meet the increased demand for ventilators during the COVID-19 pandemic, teams around the world have created low-cost, stop gap solutions to be used as a last resort. These projects were consolidated and evaluated by Dr. Robert Read and teams of volunteers at Public Invention and EndCoronaVirus.org in a public spreadsheet entitled "Analysis of Open Source COVID-19 Pandemic Ventilator Projects."
At Engineers without Borders, we felt the need to re-evaluate the clinical merit of these projects. The good intentions behind these projects are not enough as ventilators can do more harm than good if necessary settings and features are not included in a design. We believe that a high score in our evaluation better reflects the merit of a design to be used for extended care and recovery of a COVID-19 patient. A description of our new scoring categories and their weights can be viewed below. The current state of the evaluation can be viewed here. The document is open for commenting in the case changes have been made to the project since last evaluation.
Re-Evaluation Criteria:
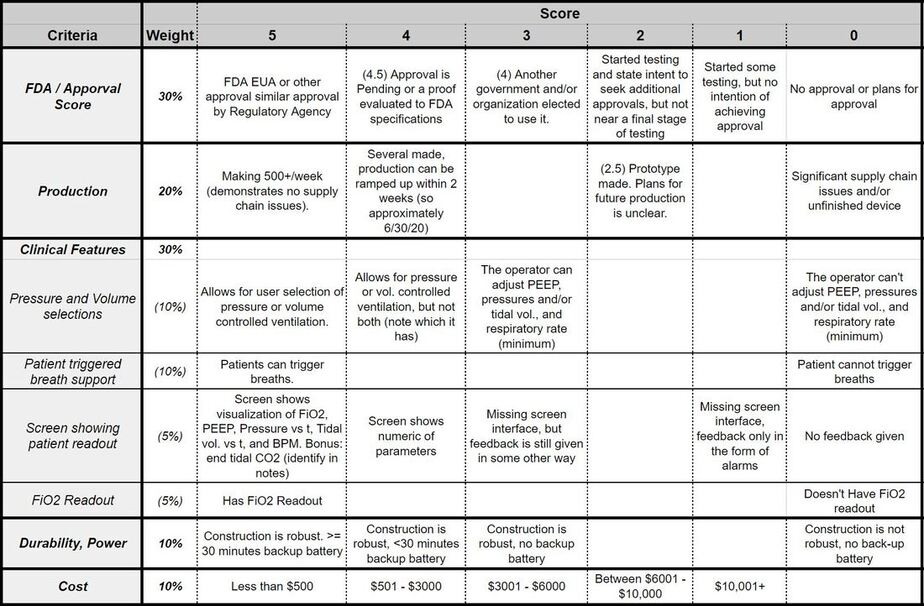
The following scoring categories were used in re-evaluating the projects on Dr. Read's Open Source Ventilator list. The scoring breakdown of each category can be viewed in the matrix beneath the criterion descriptions.
1. Approval by FDA or Other Regulatory Body (30% weight): FDA or FDA EUA (Emergency Use Approval) or similar approval from other countries is an indication that a satisfactory level of testing has been achieved. If no regulatory approval has been obtained, the testing documentation must be made available for evaluation.
2. Clinical Features (30% weight): There are many features that are necessary to ensure the safety of the patient during ventilation. We've broken these down into four subcategories:
- Pressure and Volume Control (10% weight): The best devices should allow for the user to select whether they want to use pressure or volume controlled ventilation. The clinician must also be able to adjust PEEP, pressures and/or tidal vol., and respiratory rate (minimum).
- Pressure-Support Ventilation (PSV)(10% weight): Also known as Patient-Triggered Ventilation, PTV is crucial for recovering patients to be weened off a ventilator. With this feature, the device is capable of noticing when a patient initiates a breathe and triggers the ventilator to support that breathe with pressure.
- FiO2 Readout (5% weight) : While the Fraction of Inspired Oxygen (FiO2) is easily controlled by an input valve on the oxygen source, it is crucial to have an oxygen sensor present to confirm how much oxygen the patient is actually receiving. Without it, breaks in the oxygen line or a faulty valve will deliver a false reading of inspired oxygen.
- Screen Interface(5% weight): Guatemalan clinicians specifically requested a device with a screen and familiar interface to display re-time feedback of patient metrics. The LCD or similar screen should show real-time FiO2, PEEP, Pressure vs time, (tidal) volume vs time, and respiratory rate (minimum). Added bonus if it can also report end tidal CO2.
3. Production (20% weight): Metrics on the team's current ability to produce devices communicates that there are no supply chain issue.
4. Durability and Power (10% weight): The device must be of durable construction for use in remote locations. Additionally, components should be rated for many, many cycles. A back-up battery is essential to sustain operation for at least 30 minutes to ensure uninterrupted care in the case of power-cuts. However, the lack of external power in the design is not detrimental as an external power source can easily be added.
5. Cost (10% weight): While we do not consider cost to be a driving force in evaluating the safety of a device for use with patients, it will still dictate how many devices can be purchased with a given budget.
Criteria Scoring:
This scoring gradient can also be viewed on the second tab of our "EWB Open Source Ventilator Evaluation."
Rapidly Developed Invasive and Non-Invasive Ventilators for Covid-19 [SAHPRA]
- In response to the anticipated shortage of medical supplies and equipment in the health care
system as a result of the outbreak of the Covid-19 pandemic, the South African Health Products
Regulatory Authority [SAHPRA] has drafted minimum requirements for the manufacture,
importation and distribution of rapidly developed invasive and non-invasive ventilators.